Product Overview
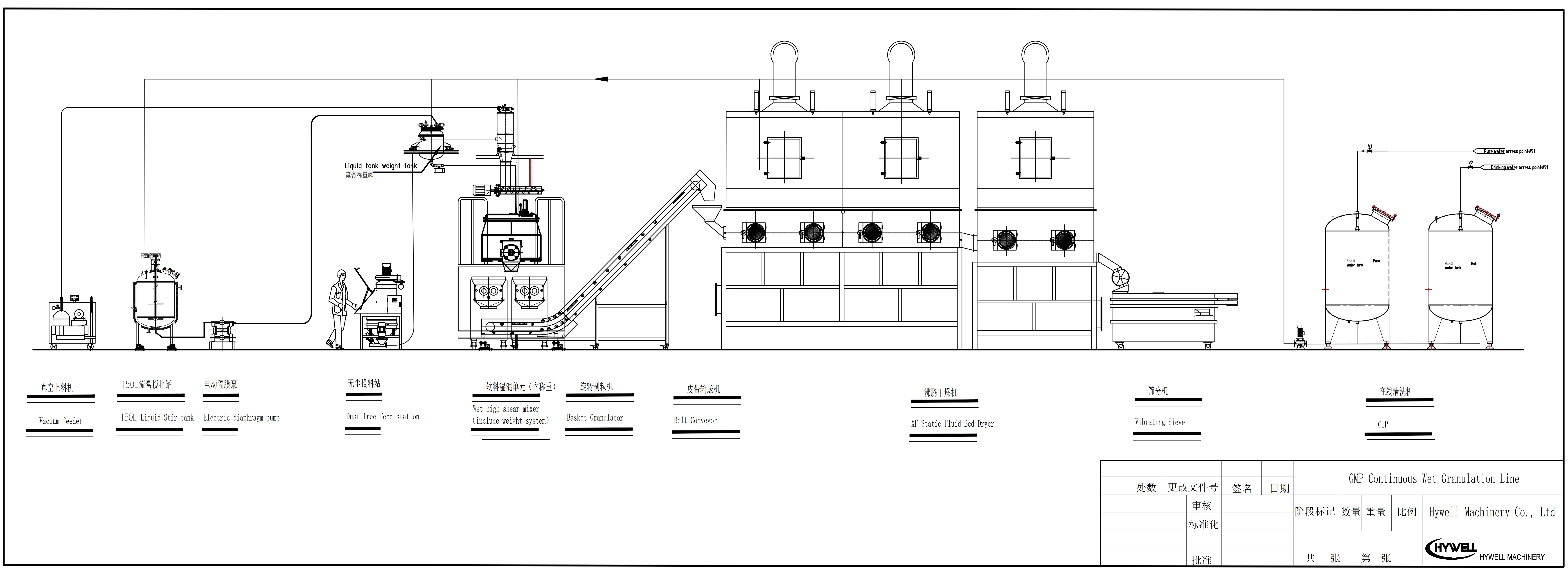
GMP Continuous Wet Granulation Line is an advanced pharmaceutical granulation production system designed for continuous manufacturing of solid dosage granules. The wet granulation system integrates a dust-free feeding station or robotic arm for feeding, vacuum feeding, a crusher (optional), a binder for mixing and weighing, a powder weighing, a high-shear mixer, a rotary granulator (basket granulator), a static fluidized bed dryer, and a vibrating screen, forming a highly efficient and fully enclosed production process.
This continuous wet granulation system is widely used in pharmaceutical factories to produce uniform, high-quality granules with excellent flowability and compressibility.
Compared with batch wet granulation systems (solid dosage forms), this continuous granulation line offers higher productivity, stable quality, and reduced labor cost, making it ideal for modern GMP production requirements. Of course, the overall investment amount is also much larger.
GMP Wet Granulation Line Process Flow
Raw Material Feeding → Vacuum Powder Conveying → Automatic Weighing & Dosing → Wet Mixing → Granulation → Drying → Sieving → Packaging → Recycling of Oversized/Undersized Particles

GMP Wet Granulation Line Video
Wet Granulator Line Key Features
Fully complies with GMP standards for pharmaceutical production
Continuous operation for higher capacity and efficiency
Closed system design to prevent dust contamination
Uniform granule size distribution (typically 16–80 mesh)
Easy integration with upstream mixing and downstream packaging
Optional CIP (Clean-In-Place) system for automatic cleaning
Reduced operator requirement (5–7 workers for the full line)
GMP Wet Granulation Line Applications
This GMP Continuous Wet Granulation Line is widely used for producing various pharmaceutical and nutraceutical granules:
Pharmaceutical Granules
• Cold & Flu Granules (Ganmaoling Granules)
• Paracetamol Granules
• Pediatric Granules
• Hpylori treatment granules
Herbal & TCM Granules
• Herbal Extract Granules
• Banlangen Granules
• Lianhua Qingwen Granules
Nutritional & Functional Granules
• Vitamin Granules
• Protein Granules
• Probiotic Granules
• ORS (Oral Rehydration Salts)

Pharma Granules

Cold & Flu Granules

Children's Medicine Granules
Proven Industry Application
This granulation line configuration is widely adopted by leading pharmaceutical companies such as China Resources Sanjiu for large-scale production of herbal and pharmaceutical granules.
The process is considered a standard wet granulation solution in China pharmaceutical industry, especially for traditional herbal medicine granules.
Main Equipment Configuration
The complete system integrates powder handling, dosing, wet granulation, drying, and screening into a fully automated pharmaceutical granulation production line. It includes the following equipment in process sequence:
1. Dust-Free Feeding Station
The dust-free feeding station is designed for safe and clean manual or automatic powder loading. It is equipped with an integrated dust collection system to prevent powder leakage, ensuring GMP compliance and improving working environment conditions in pharmaceutical production.
2. Vacuum Feeding System
The vacuum feeding system automatically transfers powder materials through negative pressure conveying, reducing manual handling and contamination risks. It improves efficiency and ensures a closed powder handling process, which is essential for modern pharmaceutical granulation line systems.
3. Powder Weighing System
The powder weighing system provides precise and automated dosing of multiple raw materials according to formulation requirements. It ensures high accuracy and repeatability, which is critical for maintaining consistent product quality in pharmaceutical wet granulation processes.
4. Binder Weighing & Preparation System
This system is used for accurate liquid binder dosing and preparation. It allows controlled addition of binder solution into the granulation process, ensuring uniform wet mass formation and improving granule strength and consistency.
5. High Shear Mixer Granulator
The high shear mixer granulator performs rapid mixing of powders and binders using high-speed impellers and choppers. It produces a uniform wet mass with excellent homogeneity, which is essential for achieving consistent granule quality in wet granulation systems.
6. Rotary Granulator (Basket Granulator)
The rotary granulator forms wet granules by forcing the wet mass through a screen. It produces a uniform particle size distribution and is especially suitable for heat-sensitive materials, making it widely used in pharmaceutical granulation production lines.
7. Static Fluid Bed Dryer
The static fluid bed dryer uses controlled hot air to efficiently dry wet granules. It ensures uniform drying, maintains product stability, and is suitable for temperature-sensitive materials in pharmaceutical and nutraceutical granulation applications.
8. Vibrating Sieve / Screening Machine
The vibrating sieve separates granules into different size fractions, removing oversized and fine particles. It ensures consistent particle size distribution, which is essential for downstream processes such as tableting or sachet packaging.

Close feeder

Weight system

Wet mixing
Technical Advantages of the GMP Granulation Production Line
1. Continuous Production Stability
Continuous production ensures stable quality and consistency, ideal for large-scale pharmaceutical granulation line operations.
2. Suitable for Heat-Sensitive Materials
Optimized wet granulation process protects heat-sensitive materials, ensuring API stability and product integrity.
3. Energy-Efficient Drying System
Fluid bed drying system reduces energy consumption while maintaining high drying efficiency in continuous granulation systems.
4. Wide Capacity Range
Scalable design supports capacities from 300 kg/h to 2500 kg/h, meeting different pharmaceutical production requirements.
5. Compatible with Solid Dosage Lines
Easily integrated with tablet and capsule production lines, forming a complete solid dosage solution.
6. High Automation & Low Labor Cost
High automation reduces manual operation, typically requiring only 5–7 operators for the entire granulation production line.
7. Fully Integrated System
Integrated powder handling, dosing, granulation, drying, and screening ensure a closed and contamination-free process.
8. Flexible for Multiple Formulations
A flexible system allows easy adjustment for pharmaceutical, nutraceutical, and herbal granules production.
9. Consistent Process Control
Stable and repeatable control ensures batch-to-batch consistency, meeting GMP pharmaceutical standards.
Why Choose Hywell?
20+ years of experience in powder processing equipment
Complete solutions for granulation production lines
Exported to 50+ countries
Strong engineering support and customization
Real GMP wet granulator production line factory
All products have CE and factory ISO certificates.
FAQ – GMP Continuous Wet Granulation Line
1. What is a GMP Continuous Wet Granulation Line?
A GMP Continuous Wet Granulation Line is a fully integrated pharmaceutical production system used to continuously produce granules by mixing, wetting, granulating, drying, and screening powders under GMP standards.
2. What are the advantages of continuous wet granulation compared to a batch granulation line?
Continuous wet granulation process line offers higher efficiency, stable product quality, reduced labor cost, and better process control compared to traditional batch granulation systems.
3. What is the typical capacity of a wet granulation production line?
The capacity of a continuous wet granulation line typically ranges from 300 kg/h to 2500 kg/h, depending on material properties and process design.
4. What materials can be processed in a pharmaceutical granulation line?
This system is suitable for pharmaceutical powders, herbal extracts, antibiotics, nutraceuticals, and other materials requiring wet granulation processing.
5. What equipment is included in a complete wet granulation system?
A standard system includes a dust-free feeding station, vacuum conveying system, powder dosing system, binder preparation unit, high shear mixer, rotary granulator, fluid bed dryer, and vibrating sieve.
6. Is the system compliant with GMP requirements?
Yes, the GMP Continuous Wet Granulation Line is designed with a closed structure, easy cleaning system, and hygienic construction to meet pharmaceutical GMP standards.
7. How automated is the granulation production line?
The system features a high level of automation, reducing manual intervention and allowing operation with only a small number of operators.
8. Can the granulation line be customized for different products?
Yes, the system can be customized based on product type, capacity, and process requirements, making it suitable for various pharmaceutical and nutraceutical applications.
9. What industries use continuous wet granulation systems?
Continuous wet granulation lines are widely used in pharmaceutical, food, chemical, and nutraceutical industries for granule production.
10. Does the system support integration with other production equipment?
Yes, the granulation line can be integrated with upstream powder mixing systems and downstream packaging or tableting equipment to form a complete production line.
Still have questions about the wet granulation line? Contact our engineers for a customized solution.
Русский
English
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
ພາສາລາວ
Bahasa Melayu
Filipino
Bahasa Indonesia
magyar
Română
Čeština
қазақ
Српски
हिन्दी
فارسی
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Dansk
اردو
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
नेपाली
Oʻzbekcha
latviešu
Azərbaycan dili
Беларуская мова
Български
ქართული
Kurdî
Кыргызча

 简体中文
简体中文




























