Introduction
Spheronization is a widely used technique in the pharmaceutical and chemical industries for the production of spherical pellets. These pellets offer several advantages over other forms of medication delivery systems. They provide controlled-release properties, improve bioavailability, and enhance patient compliance.
Understanding Spheronization
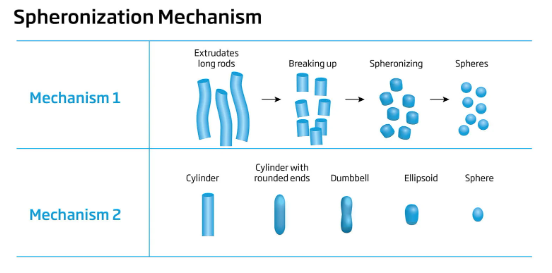
Spheronization involves the transformation of fine particles into spherical pellets through a series of mechanical and wet granulation processes. The process begins with wet granulation, where the active pharmaceutical ingredient (API) and excipients are mixed with a binder solution to form a wet mass. This wet mass is then passed through an extruder, which shapes the mass into cylindrical extrudates.
Advantages of Spherical Pellets
Spherical pellets offer several advantages in pharmaceutical formulations. Firstly, their uniform size and shape provide consistent drug release, ensuring predictable therapeutic outcomes. Secondly, the increased surface area of the pellets allows for efficient drug absorption. Lastly, the pellets can be coated to modify drug release profiles, enabling controlled and targeted drug delivery.
Materials and Equipment Required
To make spherical pellets by spheronization, you will need the following materials and equipment:
Active pharmaceutical ingredient (API)
Excipients
Binder solution
Mixer
Extruder
Spheronizer
Dryer
Sieves
Coating equipment (optional)
How do the Centrifugal Spheronizers Works Video?
Wet Mixing
In this step, the API and excipients are blended together in a mixer. The binder solution is added gradually to form a wet mass. the mixer often uses the high shear mixer, The wet mass should have a suitable consistency for extrusion.
Extrusion
The wet mass is then passed through an extruder, which shapes the mass into cylindrical extrudates. The extruder consists of a barrel and a screw that forces the wet mass through a die plate. The extrudates are cut into uniform lengths as they emerge from the die. the wet granulator mainly uses the YK oscillating granulator or basket granulator.
Centrifugal Spheronizer
The extrudates are transferred to a spheronizer, a rotating chamber equipped with friction plates. As the extrudates move within the spheronizer, the friction plates generate shear forces that round the extrudates into spherical pellets. The spheronization process usually takes around 5 to 15 minutes, depending on the desired pellet size.
Drying
After spheronization, the spherical pellets are transferred to a dryer to remove excess moisture. The drying process is crucial to ensure the stability and integrity of the pellets. The pellets are dried at a controlled temperature and humidity until they reach the desired moisture content.
Final Processing
Once the pellets are thoroughly dried, they undergo final processing steps. This may include sieving to remove any oversized or undersized pellets and achieve a uniform size distribution. Additionally, if desired, the pellets can be coated with a protective layer or modified-release coating to further enhance their functionality.
Quality Control Measures
Throughout the entire process of making spherical pellets by spheronization, strict quality control measures are implemented. This includes testing the raw materials for their suitability, monitoring the granulation parameters, conducting in-process checks, and performing final product analysis. Quality control ensures that the pellets meet the required standards of uniformity, strength, and drug content.
Video of Spherical Pellets by Spheronization Process
Applications of Spherical Pellets
Spherical pellets find applications in various pharmaceutical and chemical formulations. Some common applications include:
Controlled-release formulations: Spherical pellets can be designed to release the drug gradually over an extended period, providing sustained therapeutic effects.
Oral dosage forms: Pellets can be filled into capsules or compressed into tablets, offering flexibility in dosing and ease of administration.
Cosmetic products: Spherical pellets are utilized in cosmetic formulations for their ability to deliver active ingredients evenly and improve product performance.
Conclusion
Spheronization is an effective technique for producing spherical pellets with desirable properties for pharmaceutical and chemical applications. The process involves wet granulation, extrusion, spheronization, drying, and final processing. The resulting pellets offer advantages such as controlled release, improved bioavailability, and patient compliance. With careful attention to quality control measures, spherical pellets can be consistently manufactured to meet the required specifications.
What is a Centrifugal Spheronizer?
A centrifugal spheronizer is a specialized machine designed to convert irregularly shaped particles into spherical ones. It achieves this transformation by subjecting the particles to centrifugal forces and other mechanical actions. The resulting spherical particles find applications in various industries, including pharmaceuticals, chemicals, and food.
Working Principle
The spheronization machine is comprised of three main components: a vertical cylinder with a discharge port, a circular "friction" plate, and a variable-speed drive train that turns the plate. The working principle of a centrifugal spheronizer involves two key processes: wet granulation and spheronization. Initially, a wet granulation process is employed to create granules by blending active ingredients with binding agents and other excipients. The wet granules are then fed into the centrifugal spheronizer, which comprises a rotating disc or drum.
As the granules enter the spheronizer, the centrifugal forces generated by the rotating disc push the granules outward, causing them to collide with each other. This collision leads to the rounding of the granules, transforming them into spherical particles. Simultaneously, the wet granules are dried through the application of heated air or gas, resulting in solidified spherical particles.
Applications in Pharmaceutical Industry
The pharmaceutical industry extensively utilizes centrifugal spheronizers for various applications. One of the primary applications is in the production of pharmaceutical pellets or microspheres. Pellets offer advantages such as controlled release of drugs, improved bioavailability, and ease of encapsulation. Additionally, the spheronization process enables the uniform coating of pellets, enhancing their functional properties.
Moreover, the pharmaceutical industry employs centrifugal sermonizers for the production of multi particulate dosage forms, such as granules and beads. These multi particulates exhibit improved drug stability, reduced risk of dose dumping, and enhanced patient compliance.
Advantages of Centrifugal Spheronizer
Centrifugal spheronization offers several advantages over traditional particle processing methods. Firstly, it enables the production of spherical particles with a high degree of uniformity, shape consistency, and narrow size distribution. This characteristic is crucial for applications that require precise control over particle properties.
Furthermore, centrifugal spheronizers offer flexibility in the selection of materials and processing parameters. They can accommodate a wide range of materials, including heat-sensitive compounds. The adjustable parameters allow customization of particle size, density, and porosity, catering to specific formulation requirements.
Limitations and Challenges
While centrifugal spheronization presents numerous benefits, it also has limitations and challenges. One of the challenges is the potential generation of fine dust during the process, which may require additional measures for containment and operator safety. Additionally, certain materials may exhibit poor spheronization behavior, requiring optimization of formulation and process parameters.
Emerging Trends in Centrifugal Spheronization
Centrifugal spheronization is a continuously evolving field with emerging trends that aim to improve efficiency, enhance particle properties, and expand application possibilities. One prominent trend is the integration of process analytical technologies (PAT) into centrifugal spheronizer systems. PAT enables real-time monitoring and control of critical process parameters, ensuring consistent product quality and reducing batch-to-batch variability.
Another emerging trend is the incorporation of advanced automation and robotics in centrifugal spheronization processes. Automated systems streamline production, minimize human intervention, and improve overall process efficiency. Robotics assist in tasks such as granule feeding, disc rotation, and product discharge, optimizing the entire spheronization process.
Furthermore, research is focused on developing innovative binding agents and excipients that enhance the spheronization process. New materials with improved binding properties and compatibility with different active ingredients are being explored. These advancements contribute to the production of high-quality spherical particles with enhanced drug release profiles and therapeutic efficacy.
Future Prospects and Innovations
The future prospects of centrifugal spheronization are promising, with ongoing research and development aimed at addressing current limitations and exploring new applications. Improved containment systems and dust control measures will ensure operator safety and minimize environmental impact. Further optimization of process parameters and equipment design will lead to higher production yields and reduced energy consumption.
Moreover, the combination of centrifugal spheronization with other technologies, such as fluid bed drying and coating, opens up new avenues for the production of functional particles with tailored properties. The integration of continuous manufacturing principles in centrifugal spheronizer systems will enable seamless and efficient production of spherical particles on a large scale.
Features and Benefits
Spheronization improves your product's performance by:
• Producing a smooth particle surface suitable for thin-layer coating
• Forming granules with a specific bio-consumable shape
• Conditioning particles to prevent the generation of dust and fines from usage or shipping
• Increasing flowability by elimination of sticking or bridging points
• Increasing the apparent bulk density up to 25% compared to extruded material
FAQs
1. Are spherical pellets only used in the pharmaceutical industry?
No, spherical pellets also find applications in the chemical industry and cosmetic formulations.
2. Can spherical pellets be customized for different drug release profiles?
Yes, the coating of spherical pellets can be modified to achieve specific drug release profiles, such as immediate release, delayed release, or sustained release.
3. Are there any limitations to spheronization?
Spheronization may have limitations when dealing with highly potent or sensitive drugs that require special handling or specific release characteristics.
4. Can spheronization be scaled up for commercial production?
Yes, spheronization can be scaled up to meet the demands of commercial production. The equipment and processes can be adapted accordingly.
5. Where can I find more information on spheronization techniques?
For more detailed information on spheronization techniques, it is recommended to refer to scientific literature, and research papers, or consult with experts in the field.
In conclusion, the process of spheronization allows for the production of spherical pellets with numerous benefits in pharmaceutical, chemical, and cosmetic applications. By following the outlined steps and implementing quality control measures, manufacturers can create high-quality spherical pellets that provide controlled release, improved drug delivery, and enhanced product performance.
Русский
English
العربية
Français
Español
Português
Deutsch
italiano
日本語
한국어
Nederlands
Tiếng Việt
ไทย
Polski
Türkçe
ພາສາລາວ
Bahasa Melayu
Filipino
Bahasa Indonesia
magyar
Română
Čeština
қазақ
Српски
हिन्दी
فارسی
Slovenčina
Slovenščina
Norsk
Svenska
українська
Ελληνικά
Suomi
Հայերեն
עברית
Dansk
اردو
বাংলা
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
नेपाली
Oʻzbekcha
latviešu
Azərbaycan dili
Беларуская мова
Български
ქართული
Kurdî
Кыргызча

 简体中文
简体中文




